The ‘Angelina Effect’ helped shine the spotlight on the BRCA genes in 2013 when actor Angelina Jolie undertook radical preventative surgery to minimise her chance of developing breast or ovarian cancer. Jolie’s mother died of ovarian cancer at the age of 56, and Angelina was confirmed as carrying the BRCA1 gene mutation. Her decision to have a double mastectomy and later remove her ovaries and fallopian tubes made world headlines, raised people’s awareness of the BRCA1 and BRCA2 gene mutations, increased the uptake of BRCA gene testing, and ultimately saved many lives.
What Do BRCA Genes Do? Why is a Mutation So Bad?
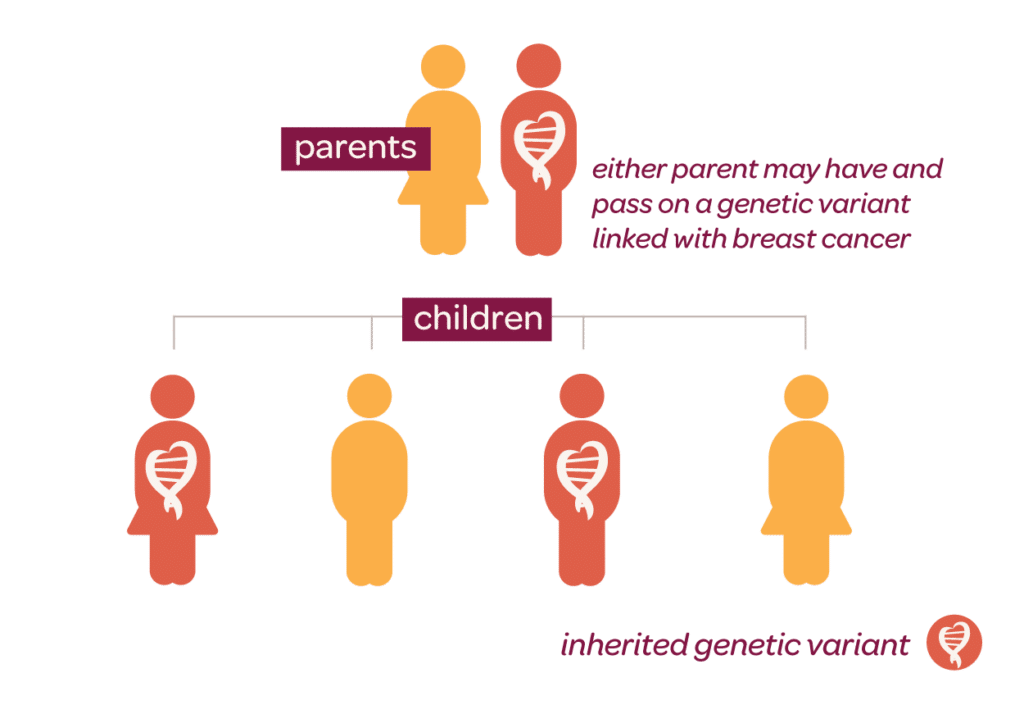
Everyone carries BRCA1 and BRCA2 genes, inheriting a copy from each parent. These genes are tumour suppressor genes linked to our DNA repair pathways, normally protecting us from getting cancer by regulating cell growth and repairing any cellular damage. Healthy BRCA genes instruct cells to make proteins that repair damage to DNA. However, if mutations occur in these genes they lose their protective effect and cells lose their ability to repair their DNA. This is when cells can start to grow and divide out of control, increasing the chances of becoming cancerous. Unfortunately mutations in these genes are associated with a much higher rate of ovarian and breast cancer. Mutations can be inherited from either parent. If one parent has the gene mutation, their child has a 50% chance of inheriting the same mutation. It is not just women that are affected by these gene mutations – faulty BRCA genes increase the risk of prostate cancer in men and pancreatic cancer in both men and women.
Angelina’s decision doesn’t seem so radical when presented with the BRCA1 carrier statistics. Carriers of the mutation have a 60 to 90% chance of developing breast cancer and a 40 to 60% chance of developing ovarian cancer in their lifetime. Around 1 in 1000 people inherit a damaged copy of the BRCA1 gene.1
Gene discovery
The link between BRCA genes and breast cancer was discovered in 1994 (BRCA stands for BReast CAncer). Doctors and scientists had known for a long time that some families had a susceptibility to certain cancers and the discovery of the BRCA genes confirmed this. Along with the gene mutation discovery came knowledge in how to develop treatments for people with BRCA cancers. Drugs called PARP inhibitors have been developed that can kill cancer cells that have the BRCA mutation. In New Zealand we have a funded medicine called olaparib that is a PARP inhibitor used in the treatment of ovarian, fallopian tube, peritoneal, BRCA-mutated breast, prostate and pancreatic cancer.2
















Community